 |
 | |
| Species: |
Rat |
| Strain/breeder: |
Wistar, Harlan Winkelmann, Germany |
| Sex: |
Male |
| Age: |
11 weeks |
| Study type: |
28 days |
| Treatment: |
Gavage |
| Animal status: |
Sacrificed at study end |
| Clinical findings: |
No abnormalities detected |
| Organ(s): |
Skin/mammary gland tissue |
Gross
finding(s): |
No abnormatities detected |
| Staining: |
H&E |
| Literature: |
 |
Andrews et al. (2002). Arch Toxicol 76: 194-202 |
|
|
 |
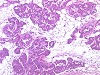
Fig. 1 control (122k)
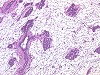
Fig. 2 treated (123k)
|
|
Abstract
Testing of endocrine active substances using an enhanced OECD test guideline (TG) 407: Experiences from studies on ethinyl estradiol
E. Hartmann and A. Freyberger
Bayer HealthCare AG, Department of Toxicologic Pathology, Wuppertal, Germany
Key words: Enhanced OECD TG 407, detection of estrogen-mediated effects, ethinyl estradiol
Groups of five male and five female Wistar rats were treated by gavage with 0.01, 0.05, and 0.2 mg/kg b.w. ethinylestradiol (EE2) for at least 28 days according to an enhanced OECD TG 407 protocol to investigate which of the current and/or additional parameters would detect effects on the endocrine system. To provide data on intra-laboratory variability, two identical studies were run in parallel. Enhancements were determination of T3, T4 and TSH, staging of the estrous cycle to ensure necropsy in diestrus, spermatology, additional organ weights (e.g., male accessory sex organs [MASO]) and additional histopathology (e.g. mammary gland, pituitary, vagina). Endocrine-mediated findings consistently observed in both studies were decreased relative weights of MASO at 0.2 mg/kg EE2, histological changes in the mammary gland (feminization) at > 0.05 mg/kg and in testes (degeneration of germinal epithelium, atrophy of Leydig cells), MASO (atrophy) and adrenals at 0.2 mg/kg in EE-treated males, estrogenization of uterus and vagina (despite of necropsy in diestrus) at > 0.01 mg/kg EE2 and changes in the ovary at 0.2 mg/kg EE2. Spermatology was insensitive. Determination of T3, T4 and TSH provided equivocal results. Doubling the group size to ten animals by combining the two studies did not increase the sensitivity of detection of endocrine-mediated effects above the level obtained by histopathological examination of groups of five animals.
 |
case index | << previous case | next case >>
|
 |